
Recently, sequencing technology and bioinformatic analyses were increasingly applied in screening genetic alterations, and further discovered potential tumor biomarkers. However, the functional role of RAD51AP1 in ESCC progression has not been studied. A number of studies have demonstrated that RAD51AP1 is overexpressed and serves as an carcinogenic role in several tumors, including ovarian cancer, lung cancer, breast cancer, as well as hepatocellular carcinoma. The silencing of RAD51AP1 leads to genomic instability and defective homologous recombination in human somatic cells. 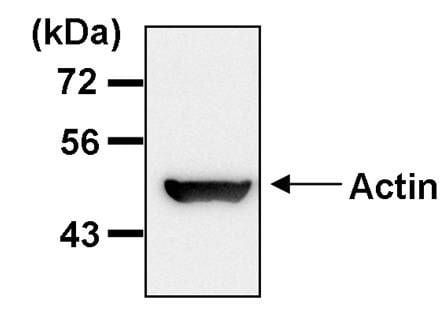
RAD51-associated protein 1 (RAD51AP1) is one kind of accessory protein for RAD51 that can vastly improve the activity of recombinase and stimulate the joint molecule formation, playing a crucial role in the repair of chromosome damage mediated by homologous recombination. A better exploitation of molecular mechanisms underlying the initiation and development of ESCC may help identify novel therapeutic targets and effective diagnostic biomarkers. It has been reported that ESCC patients has an overall 5-year survival rate of under 30%. Despite continuous improvements in therapeutic method, the survival time of patients with esophageal cancer remains unsatisfied because of late diagnosis and rapid metastasis. One of the main histological subtypes is esophageal squamous cell carcinoma (ESCC), which accounts for over 90% of the Chinese esophageal cancer patients. Conclusionsįinally, our results demonstrated that RAD51AP1 silencing significantly inhibited cell proliferation and invasion in ESCC, thereby highlighting its potential as a novel target for ESCC treatment.Įsophageal cancer represents one of the most common malignancies of upper digestive tract, ranking as the 6th leading cause of cancer-related mortalities all over the world. Transwell assay revealed knockdown of RAD51AP1 suppressed the migration and invasion of ESCC cells. Flow cytometry analysis indicated knockdown of RAD51AP1 induced cell cycle arrest and apoptosis in ESCC cells. 
Furthermore, CCK-8 and colony formation assays showed knockdown of RAD51AP1 inhibited the proliferation of ESCC cells. RAD51AP1 expression was positively associated with the enrichment of Th2 cells and T helper cells. High expression of RAD51AP1 was associated with worse survival in ESCC patients. 
RAD51AP1 expression was significantly upregulated in ESCC tissues compared with normal tissues by using The Cancer Genome Atlas (TCGA) database. The effects of RAD51AP1 on proliferation, apoptosis, migration and invasion of ESCC cells were determined by in vitro functional assays. We selected RAD51-associated protein 1 (RAD51AP1) for further research because of its poor prognosis and it has not been sufficiently studied in ESCC. Nine hub genes were identified using protein–protein interaction (PPI) network and Cytoscape software. Gene Ontology (GO) and Kyoto Encyclopedia of Genes (KEGG) pathway analyses were performed to predict the potential functions of DEGs. The overlapping differentially expressed genes (DEGs) were analyzed by GEO2R tool. Three microarray datasets were obtained from the Gene Expression Omnibus (GEO) database. This study aimed to identify potential hub gene associated with the pathophysiology of ESCC through bioinformatics analysis and experiment validation. Esophageal cancer is a common malignant tumor of digestive tract with esophageal squamous cell carcinoma (ESCC) being the main histological subtype.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
